|
The spatial distribution of electrons occupying each of these. For second period elements such as carbon, nitrogen and oxygen, these orbitals have been designated 2 s, 2p x, 2p y & 2p z. 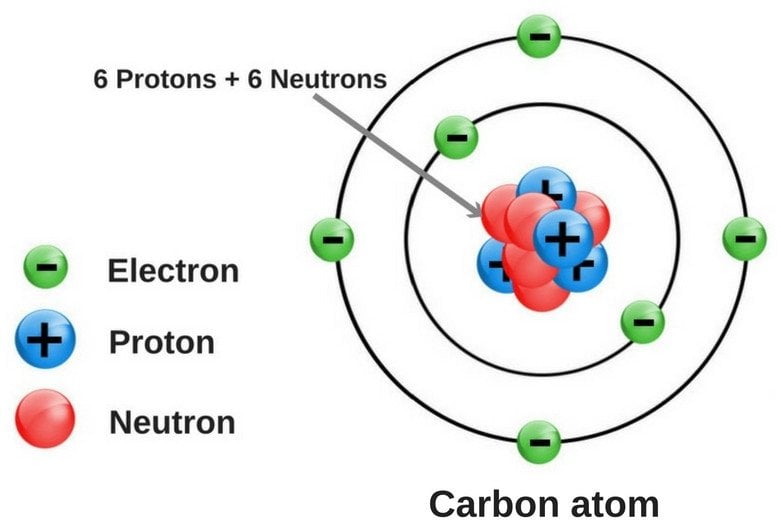
There are a number of pure forms of this element including graphite, diamond, fullerenes and graphene. It is necessary to draw structural formulas for organic compounds because in most cases a molecular formula does not uniquely represent a single compound. However, it's easy to determine the configuration of electrons for heavier elements by making a chart. The three crowns represent the three major forms of the element in nature and carbon’s status as ‘King of the Elements’ in the periodic table. If there are more electrons than protons, the ion has a negative charge and is called an anion.Įlements are shown from atomic number 1 (hydrogen) up to 94 (plutonium). Although widely distributed in nature, carbon is not particularly plentifulit makes up only about 0. Carbon is among the elements known in pure form by ancient mankind. carbon (C), nonmetallic chemical element in Group 14 (IVa) of the periodic table. The element symbol for atomic number 6 is C. If there are more protons than electrons, an atomic ion has a positive charge and is called a cation. The element name carbon comes from the Latin word carbo, which means coal. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons-or not.Īn ion of an atom is one in which the number of protons and electrons is not the same. Remember, a neutral atom contains the same number of protons and electrons. A molecule of water always contains one atom of oxygen and two atoms of hydrogen. 
The smallest particle of a compound is called a molecule. The upper right side shows the number of electrons in a neutral atom. A compound is a substance that consists of two or more elements.A compound has a unique composition that is always the same. The element atomic number and name are listed in the upper left. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 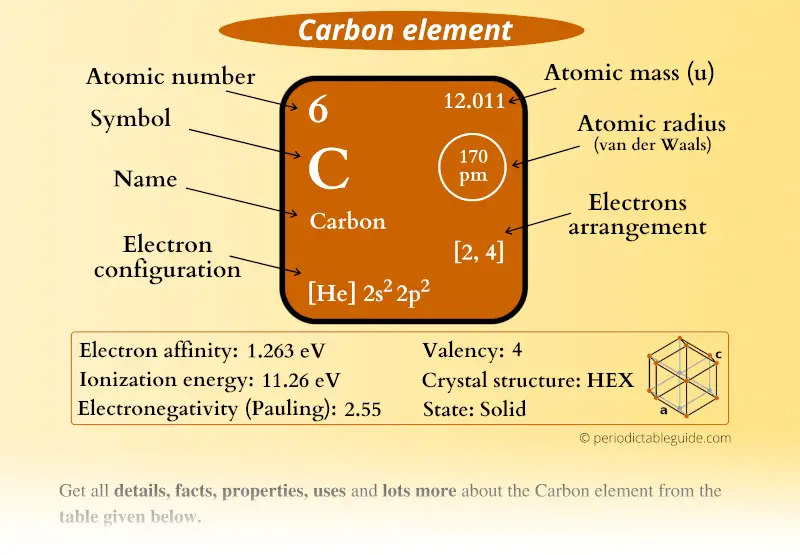
The electron shells are shown, moving outward from the nucleus. Here are electron shell atom diagrams for the elements, ordered by increasing atomic number.įor each electron shell atom diagram, the element symbol is listed in the nucleus. For that, we have electron shell diagrams. It's easier to understand electron configuration and valence if you can actually see the electrons surrounding atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
